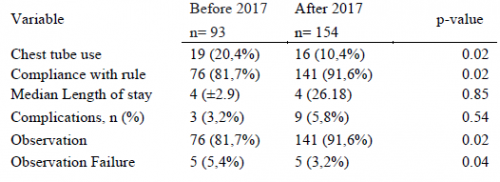
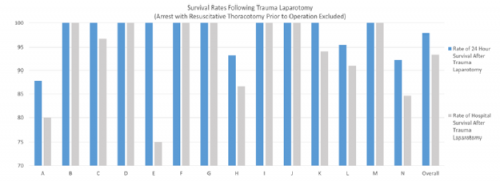
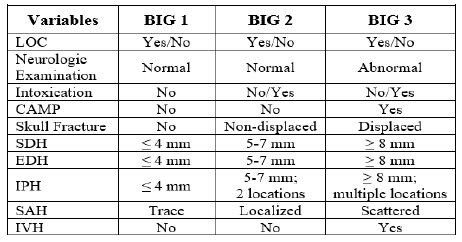
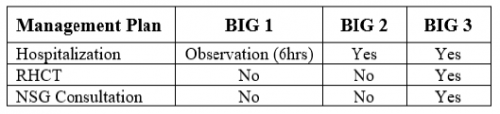
Aren’t these embarrassing? A referring center sends you a patient with the idea that they will be evaluated and admitted to your hospital. But it doesn’t work out that way. The patient is seen, possibly by a surgical specialist, bandaged up, and then sent home. Probably to one that is quite a few miles away. Not only is this a nuisance for the patient and an embarrassment for the sending center, it may use resources at the trauma center that are already tight.
Transfer patients who are seen and discharged are another form of “ultimate overtriage.” In this case, the incorrect triage takes place at the outside hospital. The trauma group in Oklahoma City reviewed their experience with these patients over a two year period. They looked exclusively at patients who were transferred in to a Level I center and then discharged.
Here are the factoids:
- A total of 2,350 patients were transferred in, and 27% were transferred home directly from the trauma bay (!)
- The three most common culprits by injury pattern were face (51%), hand (31%), isolated ortho injury (9%)
- A third of these patients required a bedside procedure, including laceration repair (53%), eye exam (24%), splinting (18%), and joint reduction (5%)
- Ten facilities accounted for 40% of the transfers
The authors concluded that the typical injuries prompting transfer are predictable. It may be possible to reduce the number of transfers by deploying telemedicine systems to push evaluations out to the referring hospitals.
Bottom line: This is quite interesting. Anyone who works in a Level I or II center is aware of this phenomenon. This abstract went a step further and quantified the specific issues involved. This center ended up discharging over 300 patients per year after transfer in. This is a tremendous drain on resources by patients who did not truly have the need for them.
The authors speculate that telemedicine evaluation may help reduce some of those transfers. This seems like an easy solution. However, it also poses a lot of issues in terms of who will actually staff the calls and how will they be compensated for their time.
There are a number of important take-aways from this abstract:
- Know your referring hospitals. In this study, there were 10 hospitals that generated an oversize number of referrals. Those are the targets / low hanging fruit. Identify them!
- Understand what their needs are. Are they frequently having issues with simple ortho injuries? Eye exams? This is what they need!
- Provide education and training to make them more comfortable. This allows you to target those hospitals with exactly the material they need and hopefully make them more self-sufficient.
This allows the higher level centers to reserve phone and/or telemedicine consultation for only the most ambiguous cases. It’s a better use of telehealth resources that may be needed, typically at night and on weekends.
Here are my questions for the presenter and authors:
- Would the common issues that were transferred and discharged be amenable to education and training at the referring centers to decrease the transfer volume?
- How have you begun to address this issue at your center?
Reference: TRAUMA TRANSFERS DISCHARGED FROM THE EMERGENCY DEPARTMENT – IS THERE A ROLE FOR TELEMEDICINE. AAST 2021, Oral abstract #63.