It happens all the time. You get that initial chest and/or pelvic xray in the resuscitation room while evaluating a blunt trauma patient. A few minutes later the tech returns with another armful of xray plates to repeat them. Why? The patient was not centered properly and part of the image is clipped.
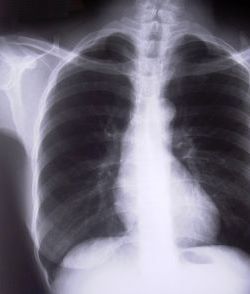
Where is the left side of the chest, and do we care?
Do you really need to go through the process of setting up again, moving the xray unit in, watching people run out of the room (if they are not wearing lead, and see my post below about how much radiation they are really exposed to), and shooting another image? The answer to the question lies in what you are looking for. Let’s address the two most common (and really the only necessary) images needed during early resuscitation of blunt trauma.
First, the chest xray. You are really looking for 3 things:
- Big air (pneumothorax)
- Big blood (hemothorax)
- Big mediastinum (hinting at aortic injury)
Look at the clipped xray above. A portion of the left chest wall is off the image. If there were a large pneumothorax on the left, would you be able to see it? What about a large hemothorax? And the mediastinum is fully included, so no problem there. So in this case, no need to repeat immediately.
The same thing goes for the pelvis. You are looking for gross disruption of the pelvic ring, especially posteriorly because this will cause you to intervene in the ED (order blood, consider wrapping the pelvis). So if parts of the edges or top and bottom are clipped, no big deal.
Bottom line: Don’t let the xray tech disrupt the team again by reflexively repeating images that are not technically perfect. See if you can use what you already have. And how do you decide if you need to repeat it later, if at all? Consider the mechanism of injury and the physical exam. Then ask yourself if there is anything you could possibly see that was not imaged the first time that would change your management in any way. If not, you don’t need it. But it certainly will irritate the radiologists!