Two days ago, I discussed getting the traditional chest x-ray routinely after chest tube insertion. The answer was yes, it is important even if it appears to be functioning correctly. But yesterday, I also showed you how the chest x-ray can lie.
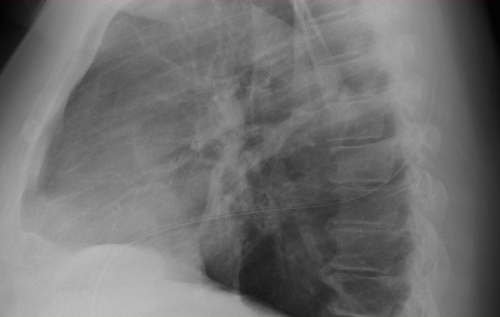
Remember this image?
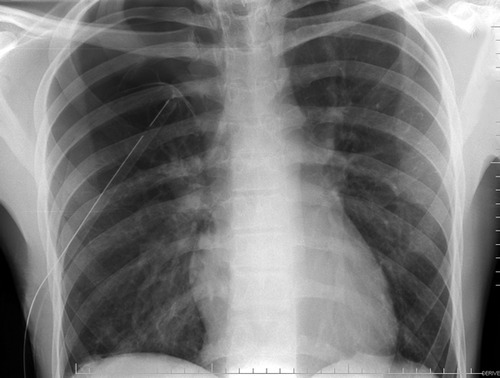
Looks perfect! But it’s a 2-D view and you don’t know where the tube is in the anterior-posterior axis. It turns out to be in the patient’s subcutaneous tissues of his back, near his scapula!
So what if this is a trauma activation patient and you are getting ready to send your patient for a chest CT shortly? Should you follow the usual dogma and still get a conventional chest x-ray prior to leaving the trauma bay?
The answer is no! Typically, your trauma activation patient should have rapid access to the CT scanner, so you won’t have to wait very long. And the additional 3-D information is very helpful in making sure the tube is placed exactly where you want it.
Bottom line: If you are planning on obtaining a chest CT anyway in your trauma patient, don’t bother with a conventional chest x-ray first to check chest tube position. But DON’T order a chest CT for this reason alone! Remember, the chest CT is only for detecting aortic injury in blunt trauma. It should not be used for diagnosing fractures, hemothorax, or pneumothorax. Or chest tube position!
Related posts: