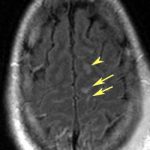
Diffuse Axonal Injury (DAI)
Trauma professionals tend to focus on the two extremes of TBI: mild concussive injury because we see so much of it, and very severe injury that we have to work so hard to keep the patient alive. Today I’ll write about the one in the middle, diffuse axonal injury (DAI). People don’t talk about it nearly as much, and it seems kind of mysterious.
DAI is also known as a shear injury, because that’s what we think happens to the brain at the time of injury. Officially, it is diagnosed when a patient remains in a coma for more than 6 hours without a demonstrable mass lesion from bleeding seen on CT. It is seen in about 15% of trauma ICU patients with head injury. Essentially the substance of the brain moves around enough to disrupt a critical mass of axonal connections that results in prolonged unconsciousness. It then takes time to try to rebuild those connections and restore consciousness and some degree of cognition. Mechanisms which result in sudden acceleration or deceleration of the brain may cause this condition, and rotational forces which spin the head suddenly seem to be even worse.
CT scan of the head frequently shows no unusual findings. On occasion, small punctate hemorrhages may be seen. These are generally bad prognostic signs, because CT is so much less sensitive in showing these compared to MRI. Here are some key points about DAI:
- If the head CT is negative,and all recreational drugs have worn off and the patient still doesn’t wake up, DAI is likely.
- MRI can confirm the diagnosis, but is not good for giving a prognosis
- Slow recovery of consciousness or failure to recover correlates with death
- Hyperglycemia and the presence of a subdural also are highly correlated with mortality
Bottom line: The diagnosis of DAI can generally be made clinically with the assistance of head CT. MRI is not very useful, unless it is needed to confirm the diagnosis. It does not predict speed or degree of recovery so is otherwise not very useful. Supportive care, avoidance of complications and early therapy and rehab are the best treatments we have to offer.
Reference: Diffuse axonal injury in patients with head injuries: an epidemiologic and prognosis study of 124 cases. J Trauma 71(4):838-846, 2011.