Blunt cardiac injury can be an enigma. Significant injuries are uncommon, and the literature on it consists of case reports and small series. The group at Scripps La Jolla has an excellent review article on the topic that is currently in press. This post will relate some of the key points in this nicely prepared article.
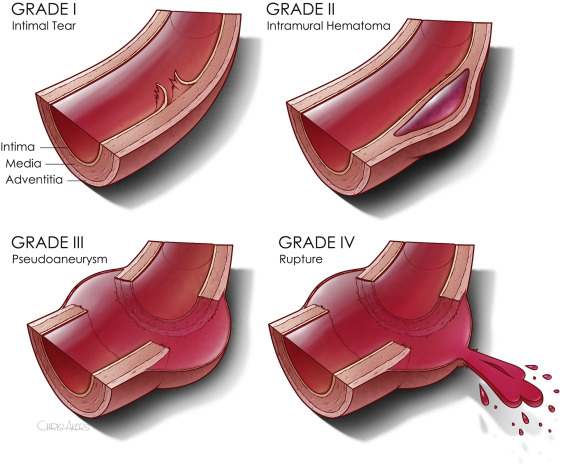
- Use the correct nomenclature. For years, many have called this condition “cardiac contusion” or “myocardial contusion.” Unfortunately, these descriptors are too specific. The proper term is “blunt cardiac injury (BCI),” which encompasses the entire gamut of injury from asymptomatic contusion to pericardial injury to cardiac rupture.
- BCI occurs more commonly than you think. If one defines BCI as any arrhythmia or cardiac enzyme elevation, it is fairly common. However, if the definition is limited to clinically significant sequelae such as potentially malignant arrhythmia or cardiac failure, the incidence is easily less than 1% in blunt trauma patients.
- Be aware of the usual mechanisms of injury. This is a condition caused by blunt trauma, with motor vehicle crashes causing half and pedestrians struck by them another one-third. Motorcycle crashes and falls caused the remaining 12%.
- Diagnosis can be challenging.
- Physical examination is usually of little help. New onset of a heart murmur may indicate a serious cardiac injury but is exceedingly rare.
- EKG evidence of a new onset arrhythmia is important, particularly bundle branch blocks, PVCs, and ST segment / T wave changes, which require further investigation.
- CPK-MB enzyme measurements are useless. Please don’t get them.
- Troponin T and Troponin I are frequently used but do not reliably predict BCI. Testing in asymptomatic patients is not helpful and may result in additional asymptomatic testing.
- Echocardiography is not indicated in asymptomatic patients with isolated enzyme elevations.
- Cardiac CT may be used to differentiate acute MI from BCI. Frequently, patients at risk are having a chest CT with contrast performed anyway.
Here is the recommended treatment algorithm:
- If BCI is possible based on mechanism of injury, follow the ATLS protocols and perform a physical exam, E-FAST, and place on EKG monitoring.
- If the patient is hemodynamically unstable, quickly identify and treat tamponade or tension pneumothorax if present. If significant arrhythmias are present, treat with appropriate medications. If heart failure is present, treat medically and evaluate for surgical problems such as valve, septum, or coronary artery injury.
- If the patient is hemodynamically stable, obtain a 12 lead EKG. If significant arrythmias are present, treat with appropriate medications. If there is organ hypoperfusion, obtain an echocardiogram. If this study reveals an effusion, a pericardial window is indicated. If the echo shows hypokinesis or structural injury, appropriate medical or surgical management should be carried out.
- Patients who have only significant arrhythmias should be admitted to a monitored bed for 24 hours. Once arrhythmias have resolved, the patent can be discharged.
- Patients with nonspecific EKG changes should have troponin levels drawn after 8 hours of observation in the ED. If elevated, admit to a monitored bed for 24 hours. Once EKG and troponin have normalized, the patent can be discharged.
- If EKG and labs are normal, may discharge home from the ED if there are no other indications for admission.
Reference: Diagnosis and Management of Blunt Cardiac Injury: What You Need to Know. J Trauma, accepted for publication. DOI: 10.1097/TA.0000000000004216