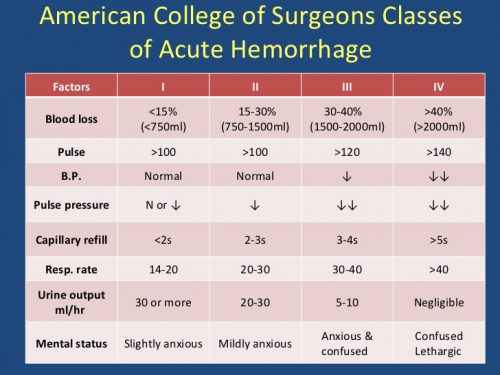
Here’s an operative tip for trauma professionals who find themselves in the OR. Heard of “lunchothorax?” I’m sure most of you haven’t. The term originated in a 1993 paper on the history of thoracoscopic surgery. It really hasn’t been written about in the context of trauma surgery, though.
Lunchothorax is an empyema caused by pleural contamination in patients with concomitant diaphragm and hollow viscus injury. This most commonly occurs with penetrating injuries to the left upper quadrant and/or left lower back. The two penetrations tend to be in close proximity (diaphragm + stomach), but may occasionally be further away (diaphragm + colon).
One of the earlier papers describing the correlation of gastric injury and empyema was written by one of my mentors, John Weigelt. Although gastric repair is usually simple and heals well, his group did note a few severe complications. Of 243 patients with this injury, 15 developed ones that were considered severe, and 10 of those were empyema! What gives?
It turns out that the combination of gastric contents and pleural space is not a good one. It’s not really clear why this is. Is it bacterial? The acid? Undigested food? I’ve seen cases with what I would consider minimal contamination go on to develop a nasty empyema. This is also borne out in a National Trauma Databank review from 2009. It looked at complications in patients with a diaphragm injury and found that a gastric injury increased the probability of empyema by 3x. Interestingly, there was no increased risk of empyema with a concomitant colon injury.
Bottom line: Lunchothorax, or empyema after even minimal contamination from a hollow viscus, is a dreaded complication of thoraco-abdominal penetrating injury. Any time the stomach and diaphragm are violated, I recommend thoroughly irrigating the chest. It’s probably a good idea for concomitant colon injury as well, but there’s less literature support.
This can be done through the diaphragm injury if it is large enough, or through a chest tube inserted separately. Most of the time, you’ll be placing the chest tube anyway because the pleural space has been violated via the abdomen. In either case, copious lavage with saline is recommended to clear all particulate material, with a few extra liters just for good measure. There’s no data on use of antibiotics, but standard perioperative coverage for the abdominal injuries should be sufficient if the lavage was properly performed.
References:
- The history of thoracoscopic surgery. Ann Thoracic Surg 56(3):610-614, 1993.
- Penetrating injuries to the stomach. SGO 172(4):298-302, 1991.
- Risk factors for empyema after diaphragmatic injury: results of a National Trauma Databank analysis. J Trauma 66(6):1672-1676, 2009.