Here’s a new pop quiz for everyone. You’re all so good I’m not giving any hints on this one:
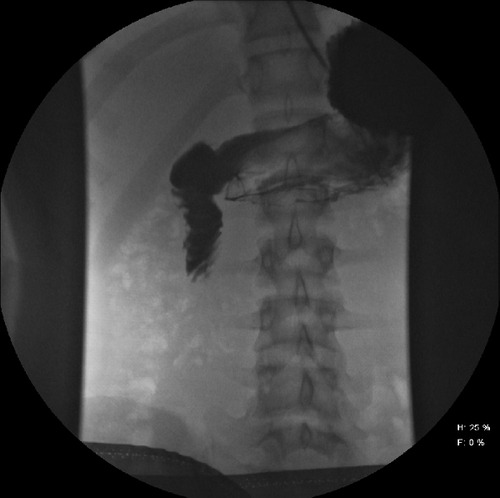
Now, the questions for you:
- What happened? Exact mechanism, please.
- What’s the pathology?
- What do you do about it?
Comment below or tweet to #TraumaPro
Here’s a new pop quiz for everyone. You’re all so good I’m not giving any hints on this one:

Now, the questions for you:
Comment below or tweet to #TraumaPro
Trauma hurts like hell. Over the years, we’ve developed quite a few ways of combating this pain. A number of drug classes have been developed to reduce it. One of the more common non-narcotic drug classes are the NSAIDs. As I’ve mentioned before, every drug has dozens of effects. Drug companies market a particular medication based on one of the predominant effects. All the others are considered side effects.
NSAIDs are not unique; they have lots of side effects as well. In 2003, several papers brought to light possible interactions between these drugs and fracture healing. Specifically, there were questions about these drugs interfering with the healing process and of increasing the number of delayed unions or nonunions. But once again, how convincing were these papers, really?
It would seem to make sense that NSAIDs could interfere with bone healing. This process relies heavily on the regulation of osteoblast and osteoclast function, which itself is regulated by prostaglandins. Since prostaglandins are synthesized by the COX enzymes, COX inhibitors like the NSAIDs should have the potential to impair this process. Indeed, animal studies in rats and rabbits seem to bear this out.
But as we have seen before, good animal studies don’t always translate well to human experience. Although a study from 2005 suggested that NSAID administration in older patients within 90 days of injury had a higher incidence of fracture nonunion, the study design was not a very good one. It is equally likely that patients who required these drugs in this age group may have been at higher risk for nonunion in the first place.
In fact, there are no large, prospective randomized studies that have explored the effect of short-term or long-term NSAID administration on fracture repair. But there have been several smaller studies that showed absolutely no effect on nonunion with short-term administration of this drug class. Yet the dogma that leads us to avoid giving these drugs persists.
Bottom line: Once again, the animal data is clear but the human data is not. Although there are theoretical concerns about their use, there is not enough solid risk:benefit information to abandon short-term NSAID use in patients who really need them. NSAIDs can and should be prescribed in patients with short-term needs and simple fractures.
References:
So you’re faced with a chest tube that “someone else” inserted, and the followup chest xray shows that the last drain hole is outside the chest. What to do?
Well, as I mentioned, there is very little written on this topic, just dogma. So here are some practical tips on avoiding or fixing this problem:
Related posts:
Yesterday I presented the problem of the malpositioned chest tube, specifically one that is not completely in the pleural space. This one is way out:
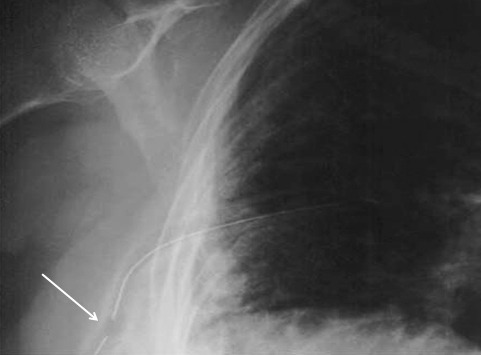
So what do the doctor books say? Well, the first thing you will discover if you try to look it up is that THERE IS NO LITERATURE ON THIS COMMON PROBLEM! There are a few papers on tubes placed in the fissure and tubes inserted into the lung parenchyma. But there are only a few mentions of tubes with holes still outside the chest.
I’ve gotten a number of comments, including “you can push them in a little”, “take it out and put in another”, and “never push them in.” Since we don’t have any science to guide us, we have to use common sense. But remember, I’ve shown you plenty of examples where something seems reasonable, but turns out to be ineffective or downright harmful.
There are three principles that guide me when I face this problem:
Monday, I’ll finish this series and provide some tips and guidelines to help manage this problem using the principles outlined above.
The November newsletter is here! Click the image below or the link at the bottom to download. This month’s topic is Trauma Mythbusters, discussing the dogma surround and the (lack of) data supporting:
Subscribers had the newsletter emailed to them on Tuesday. If you want to subscribe (and download back issues), click here.