Even though it’s called trauma surgery, the operative experience in trauma tends to be somewhat limited. This is due mostly to the fact that most trauma centers see predominantly blunt trauma. Yes, there are hospitals around the world where the penetrating injury load remains high and there is operative experience aplenty.
But in the US, the vast majority of trauma centers see mostly blunt trauma. Surgical residents in the US are required to log 10 operative and 20 nonoperative cases to successfully meet residency completion requirements. And blunt trauma is tending to get less and less operative in nature. A good example is the evolution of blunt solid organ injury to mostly nonoperative management.
So what is happening with surgical resident operative trauma experience? And has there been any impact from the work hour restrictions that have gone into effect in the US? A study from Harborview, Denver Health and Seattle Children’s looked at the ACGME operative logs for surgical residents annually from 1989 to 2010. They combined the data into 5 year blocks, with the last two having work hour restrictions in place.
Some interesting findings:
- Overall mean caseload of major cases (all types) remained steady at about 925 per resident
- Mean trauma operative caseload decreased from 76 to 39 (beginning of work hour restrictions)
- Mean trauma operative caseload remained steady at 39 for the 7 years in which work hour restrictions were in effect
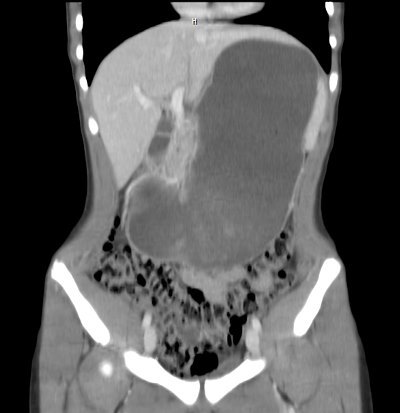
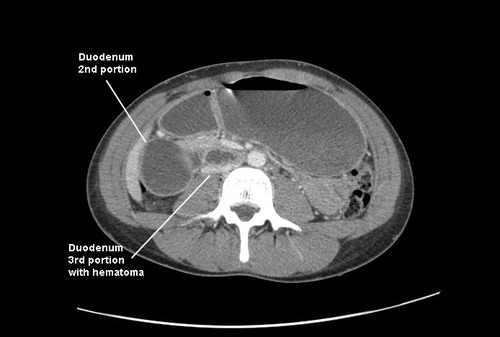
- The number of intra-abdominal trauma operations decreased from 31 to 17, and the number of liver/spleen operations decreased from 5 and 4 to 3 and 2
Bottom line: Resident trauma operative experience has declined and stabilized in the US. This is due to the evolution of our management of blunt trauma. Unfortunately, this decline will reflect on how well prepared surgeons at outlying hospitals are, and in the quality of emergency surgery they may provide. The impact will be felt most by seriously injured patients who cannot be taken to a high level trauma center initially. We need creative solutions to address this issue, such as mini-clerkships in trauma or structured experiences at high level trauma centers for surgeons in outlying hospitals.
Related post: ED at the busiest hospital in the world!
Reference: ACGME case logs: surgery resident experience in operative trauma for two decades. J Trauma 73(6):1500-1506, 2012.