Fasciotomy is the definitive management for compartment syndrome. But by definition, once you make the incision things are going to gape apart. If they don’t, hmmm, there probably wasn’t a compartment syndrome in the first place.
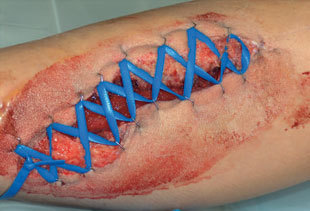
That’s the easy part. Now, how do you make the wound edges come together and achieve some kind of cosmetic result? Historically, a variety of techniques have been used. They include leaving it open to granulate, brute force sutures, progressive closures, and more recently, the VAC suction dressing.
This latter technique has really caught on, and there are a number of benefits. First, the suction can reduce tissue edema, which may facilitate quicker closure. Another big advantage is that this dressing can be changed every 3 days, as opposed to daily (or more) for conventional dressings.
The downsides: cost, and the fact that some people in the US don’t have insurance that covers home use of this device. This may drive up costs by increasing hospital length of stay.
But is it better than the other closure methods? A recently published paper from an orthopaedic and plastic surgical group in Greece details a randomized, prospective study comparing VAC assisted closure versus the shoelace technique.
Here are the factoids:
- 50 patients with 82 leg fasciotomy wounds were randomized over 5 years
- The VAC group had the device applied 3-6 days postop with a pressure of -125 torr. They were changed every 3 days.
- The shoelace group had the bands applied at the end of the operation. Tightening began 4-6 days postop and was then performed daily.
- Time to closure with the VAC was 19 days vs 15 days for shoelaces. This was a significant difference.
- Skin grafts were required to complete the closure in 6 VAC cases, but in none of the shoelace patients
- There were 6 wound infections in the VAC group vs 4 in the shoelace group (NS)
- Average daily cost in the VAC group was 135 euro, but only 14 euro for shoelaces
- The cost to add a skin graft in the VAC group added substantial additional expense
Bottom line: This is a nice comparison of two techniques that try to solve the wound closure problem using two different methods. The VAC reduces edema but does not shrink the wound, while shoelaces stretch the skin to close the wound but do nothing about edema. The VAC is slower and more expensive, and frequently requires an additional (and expensive) skin graft. Shoelaces are quicker and cheap. What to do? It would appear that wound shrinking methods are preferred. However, if edema is significant, apply a VAC first. Then switch to shoelaces once the edema has subsided for faster (and graft-less) closure.
Related post:
Reference: Wound closure of leg fasciotomy: Comparison of vacuum-assisted closure versus shoelace technique. A randomised study. Injury 45(5):890-893, 2014.

