What Next? An Inkjet Printer for Skin?
Everyone is familiar with inkjet printer technology. You’ve probably got one in your house for printing 2D page images from your computer. Engineers have already taken this one step further and created 3D printers that print objects from computer aided design (CAD) files. Instead of shooting tiny dots of ink from a cartridge, they squirt out tiny dots of molten plastic.
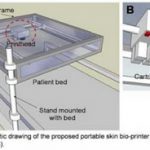
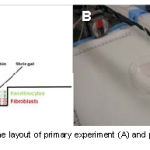
This same technology is poised to change the way we do things in medicine. James Yoo and colleagues from Wake Forest have designed a printer that can print skin. This unit has been redesigned from earlier versions and now uses a laser to scan the contours of the area to be grafted. It then prints a skin graft over the area using different layers of cells.
The Department of Defense is funding this work, which has amazing implications for the battlefield and for disaster areas. Imagine being able to print a skin graft onto a wounded soldier or civilian to reduce fluid loss and decrease infections. In these applications, cartridges of skin cells are more easily transported than freezers of cadaver skin. However, these grafts would be temporary, just like cadaver or pig skin, because the cells would be from unmatched donors. But ultimately, we should be able to prepare cartridges from our own cells for long lasting grafts.
The Wake Forest group is successfully printing 10x10cm grafts onto pigs right now. But think of the broader implications of this technology. Other groups are looking at using 3D printer technology to squirt a variety of cell types to create complete organs. This could eventually revolutionize transplant technology as we know it!
References
- In Situ Bioprinting of the Skin for Burns. Binder, Yoo et al. Presented at the American College of Surgeons Clinical Congress, October 5,2010, Washington DC.
- Presentation at the American Association for the Advancement of Science, February 16-20 2011, Vancouver BC, Canada.