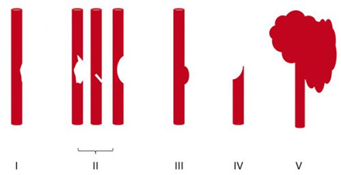
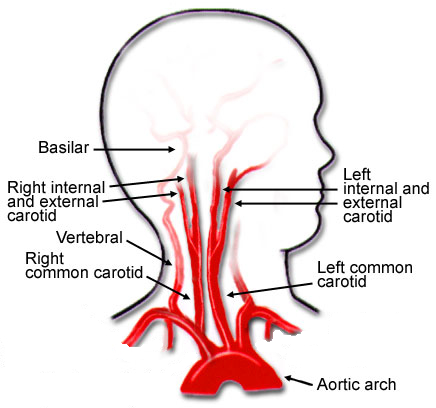
Several days ago, in my post on “How Common Is BCVI?” I mentioned a paper recognizing the increasing incidence of BCVI in pediatric patients and the very high stroke rate (37%) and death rate (13%). These numbers are very concerning!
Previous work shows that the Memphis and Denver criteria are not very sensitive in adults. This has led many trauma centers to add CT angiography of the neck automatically in patients with a high-energy mechanism. But what about kids? Are these screening criteria any better?
A consortium of children’s hospital trauma centers has a paper currently in press that evaluated both the Memphis and Denver criteria in children under age 15. It was a four-year prospective, observational study of children with head, face, or neck injuries. Although the Memphis criteria were specifically used in the study, data for applying the original and expanded Denver criteria, EAST practice management guideline criteria, Utah score, and McGovern score were also collected. The last two are pediatric-specific criteria.
Any child who met at least one of the Memphis criteria received a CTA or MRA of the head and neck. In addition, all children with head, face, or neck injuries received a follow-up evaluation two weeks after discharge. This was designed to capture any evidence of BCVI in those who did not meet Memphis criteria and hence had no CTA/MRA. Patients who missed this evaluation or had other missing data were excluded from the analysis.
Here are the factoids:
- A total of 2,284 children met the criteria for enrollment; nearly one-third were excluded due to no imaging/follow-up or missing data (!)
- There were 24 BCVI diagnosed (1.6%)
- Diagnostic accuracy of the various screening criteria were:
| Criteria | Sens | Spec | PPV | NPV | # CTA to detect one BCVI |
| Memphis | 92 | 71 | 5 | 100 | 20 |
| Denver | 73 | 88 | 9 | 100 | 11 |
| Expanded Denver | 88 | 64 | 4 | 100 | 25 |
| EAST | 79 | 83 | 7 | 100 | 14 |
| Utah | 49 | 96 | 16 | 99 | 7 |
| McGovern | 75 | 90 | 11 | 100 | 9 |
The Memphis criteria had the highest sensitivity and would have missed the fewest BCVI. The pediatric-specific Utah score had the highest specificity but would have missed more than half of the injuries. The authors recommend refining the Memphis criteria to improve its specificity while maintaining its high sensitivity.
Bottom line: As with adults, we struggle with systematically identifying BCVI. All screening systems leave something to be desired. It’s not practical or prudent to treat children the same as adults and just liberalize the use of CTA. Substituting MRA is not practical because this requires sedation and/or intubation in the younger age groups.
Of interest in this study, the overall incidence was higher (1.6% here vs. less than 0.5% in my previous post). This is probably due to the fact that there was a significant effort to identify criteria for angiography, and follow-up was provided to detect occult injuries.
This paper adds to the previous work I cited describing how important it is to detect this injury. The current research demonstrates that the Memphis criteria are the best we have for pediatric patients at this time. But it clearly shows the need for a better tool.
But until one is developed, a best practice would be to use the Memphis criteria to screen any pediatric patient with head, neck, or facial trauma due to a high-energy blunt mechanism. Then select CTA or MRA after conferring with your pediatrics and radiology teams.
Reference: Diagnostic accuracy of screening tools for pediatric blunt cerebrovascular injury: An ATOMAC multicenter study. Journal of Trauma, publish ahead of print, DOI: 10.1097/TA.0000000000003888.