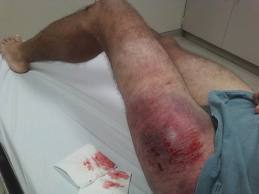
In the last two posts, I’ve examined the phenomenon of “delayed diagnosis” or missed injury. I believe that there are only two fundamental reasons why this occurs:
- Insufficient diagnostic technique – A good physical exam and/or specific diagnostic techniques were not performed. Or rarely, the injury cannot be readily detected by existing techniques and technology. The former is usually the real problem and may be an issue with either the physical exam completeness and/or technique, or judgment used to obtain the appropriate diagnostic test. Example 1: a penetrating injury to the back is missed because the patient is not logrolled to examine this area. Example 2: a spine fracture is missed in an elderly patient with a fall from standing because the back pain found on physical exam is evaluated only with conventional imaging of the spine, not CT.
- Failure to recognize the injury – The injury was actually identified on a test but was not appreciated by the clinician. Example 1: the radiologist may not have appreciated and reported out a subtle anomaly in the cervical spine imaging. Example 2: you fail to check your patient’s lab tests and miss a sudden spike in serum amylase or lipase the day after your patient was kicked in the epigastrium by a horse.
So what can you do to avoid this potential problem? Here are some tips:
- Admit that it can really happen to you. If the missed injury rate at your center is off the low end of the bell curve (< 5%), then you are either really good or really blind. You’d better take a close look at your performance improvement process because you may be fooling yourself.
- Adopt a firm definition of “delayed diagnosis.” Basically, you need a time frame after which a new diagnosis is considered “delayed.” It should be a reasonable time interval after the patient has left the ED. If it’s too short an interval (e.g. once they leave the ED), your number will be unnecessarily high. If it’s too long (days and days later), then significant morbidity may occur that you don’t account for. Most centers have adopted 24, 36, or 48 hours after patient arrival.
- Implement a tertiary survey process. This is a complete physical re-examination followed by a review of all diagnostic studies (lab and radiology) that have been performed. This exam needs to be dated and timed to ensure that it is performed within the time frame noted above. If a new finding is discovered on the tertiary survey, it is not considered a delayed diagnosis. If found after the survey (or after the pre-determined time interval), it is and must be entered into your performance improvement process.
- Be paranoid. I hate the phrase, “maintain a high index of suspicion” because it’s meaningless. It’s like those stupid “start seeing motorcycles” bumper stickers. You can’t see what you can’t see. But you can be suspicious all the time, constantly looking for the inevitable clinical surprises of trauma care.