In my last post, I described an elderly pedestrian struck by a car. During the trauma activation, routine chest and pelvic x-rays were obtained. Here was the pelvic image:
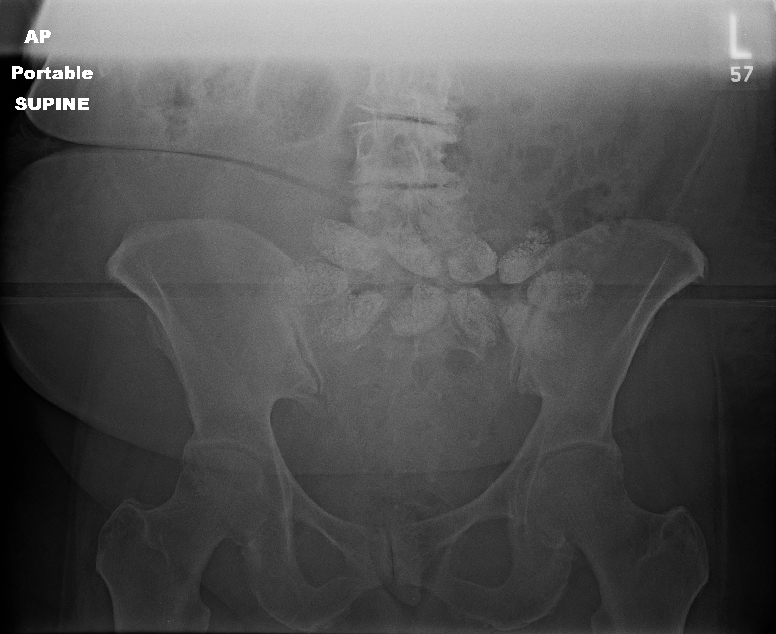
Note the odd oval densities across the center of the x-ray. What the heck? What are they?
There are two choices: they are either inside or outside the patient. We had already removed her clothes, so it wasn’t something she was carrying. And if it was inside, we would be able to identify it on the CT scan we had ordered.
But in this case, the x-ray was done early in the secondary survey. Specifically, we did it before we rolled our patient and examined her back. When we did, here is what we found:
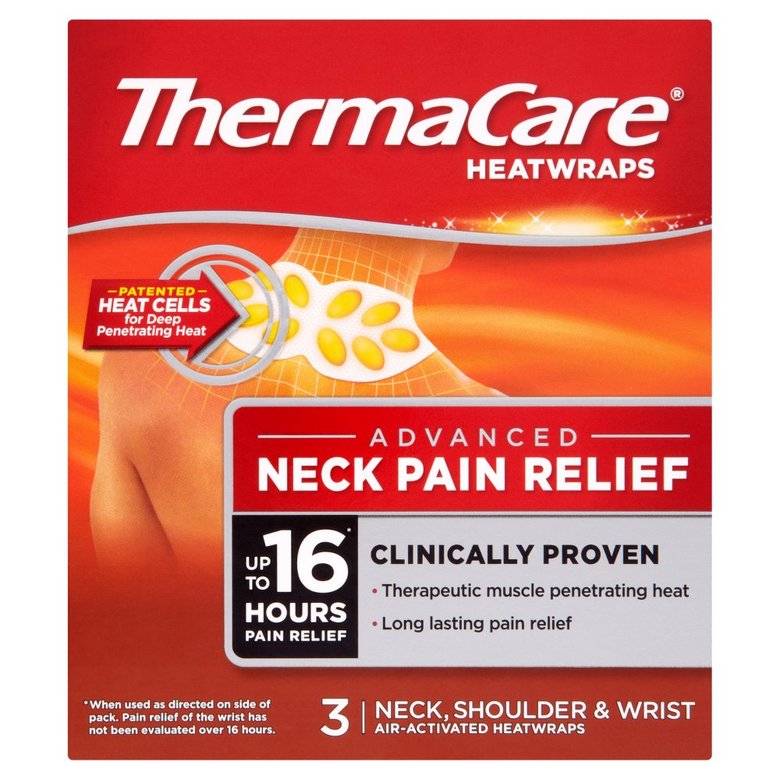
Only it wasn’t in the box. Or on her neck. This one was stuck on her lower back, but not in her clothes. She was suffering from lower back pain, and applied one of these on a daily basis for comfort. We had not rolled the patient prior to the pelvic x-ray.
The pods on these thermal wraps contain a mixture of iron, sodium chloride, sodium thiosulfate, water, charcoal, and sodium polyacrylate that heat up when removed from their package and exposed to oxygen. The iron renders it somewhat radio-opaque, hence their appearance on the x-ray. We did peel it off prior to CT since it would probably create a significant amount of scatter which would degrade the image.
Should we have waited a few more minutes to get the image until we had rolled and examined the back? This is a judgment call. Since our trauma team moves quickly, we are typically ready to head to the scanner in 15 minutes. In order to improve overall CT scan throughput, we have adopted a 5-minute advance notice policy. To accomplish this, we don’t want to wait until the very end of the resuscitation to get x-rays. That would end up slowing down our process.
You may feel differently about the timing of the images, or you may have a different method of sequencing your CT scanner. Whatever works best for you. But remember, all trauma patients need to be completely undressed and all of their surfaces, nooks, and crannies inspected before they leave the emergency department!

