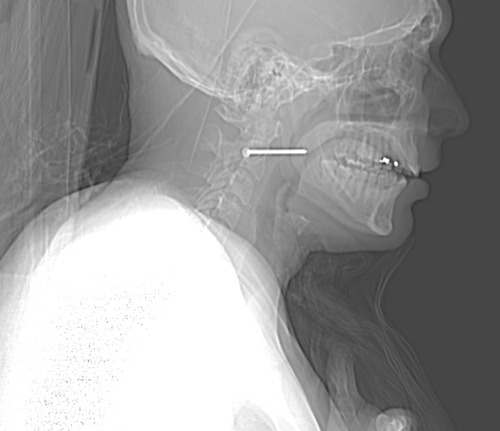
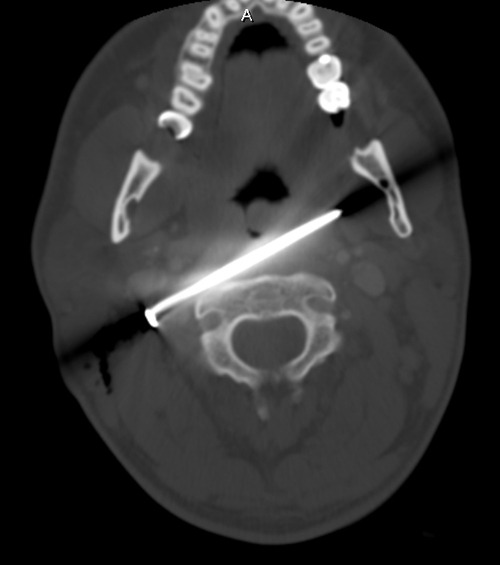
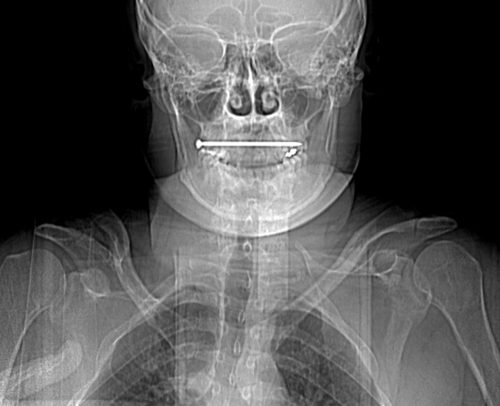
Here’s a post from my archive describing a different way to remove the foreign body. This is the technique I used, instead of the standard neck incision. The final incision was just a slight extension of the puncture wound, measuring only 1cm. I was able to grasp the head and pull it out without difficulty. The surprising thing to me was the amount of force I needed to apply to actually pull it out! No bleeding, no problems. The patient was observed for 24 hours and discharged home. He had no complications.
A Cool Way To Remove Embedded Foreign Bodies
Many of us have had the experience of digging into bloody tissue for long periods of time trying to locate the object, even with fluoroscopy. Well, there’s a better way of doing this.
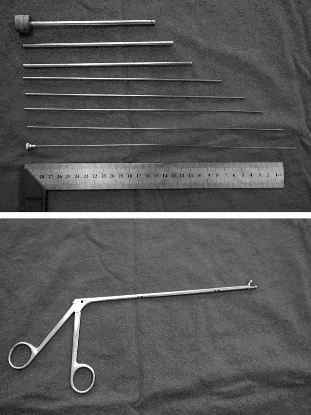
A group in China described a technique using a fancy form of needle localization. They employed a set of instruments normally used for lumbar diskectomy (see photo). This set includes a long 18 Ga needle with a removable hub, several dilators and an outer cannula with a 5.8mm diameter. A pair of 3.8mm grasping forceps is also used.
The foreign body is located using a C-arm fluoroscopy unit and the best approach is planned. The 18 Ga needle is then inserted using fluoro until it touches the object. The hub is removed and dilators are inserted over the needle, one after the other. The outer cannula is then placed over them, and the needle and dilators are then removed. The cannula is manipulated until the foreign body (or a part of it) is located within the cannula. It is then grasped and removed, along with the cannula if needed. If the object is too large to enter the cannula, the cannula is pulled back slightly and the grasper introduced past the end of it to grip and remove the foreign body.
The writers shared the details of 76 patients who had a total of 251 foreign bodies removed over a 6 year period. The depth varied from 2.5 to 8.5cm. Procedure time ranged from 8 to 15 minutes, and fluoro exposure varied from 1 to 4 minutes. Success rate was 100% (all foreign bodies were removed) and there were no complications.
Bottom line: This is a very slick technique that promises to dramatically increase the success rate and decrease complications from removing foreign bodies. The amount of time spent is much less than the brute force technique, as is the amount of soft tissue trauma. Large objects that cannot be grasped with these forceps cannot be removed with this method. Although I am a little concerned that the authors’ results were so perfect, it’s certainly worth a try!
Reference: Percutaneous extraction of deeply-embedded radiopaque foreign bodies using a less-invasive technique under image guidance. J Trauma 72(1):302-305, 2012.