Great questions, and I’d love to answer in detail. Please contact me by email (see my profile at the side of the page) so we can chat!
All posts by The Trauma Pro
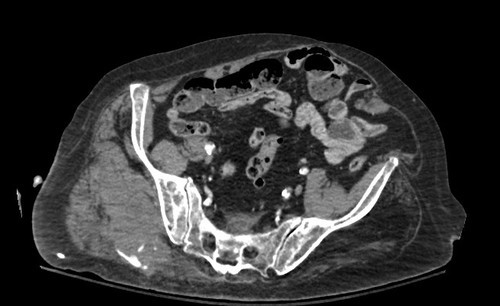
Pop Quiz! Hints
So far, no correct answers to all three questions from yesterday’s pop quiz! Given the scan below, I want to see if you can figure out:
- Mechanism of injury
- Age range
- Diagnosis
And bonus points for the correct answer on how to treat the problem!
Hint: You can tell the age by the appearance of the blood vessels in the pelvis. And the bright areas outside the pelvis may not be calcium. This should give you enough information to figure out the rest.
Tweet or comment your results!
Pop Quiz! You Figure It Out
Here’s a typical barebones clinical problem for you:
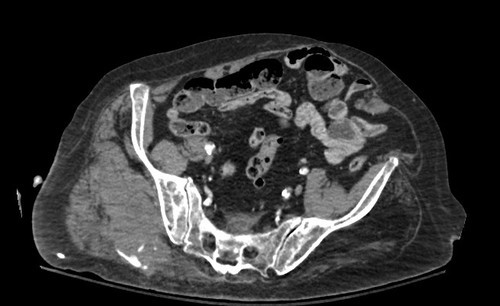
Given the usual statistical probabilities, tell me these three things:
- Mechanism of injury
- Age range
- Diagnosis
Please tweet to @RegionsTrauma or leave comments with your answers! More info tomorrow!
CT Cystography For Bladder Trauma
Bladder injury after blunt trauma is relatively uncommon, but needs to be identified promptly. Nearly every patient (97%+) with a bladder injury will have hematuria that is visible to the naked eye. This should prompt the trauma professional to obtain a CT of the abdomen/pelvis and a CT cystogram.
The CT of the abdomen and pelvis will identify any renal or ureteral (extremely rare!) source for the hematuria. The CT cystogram will demonstrate a bladder injury, but only if done properly!
During most trauma CT scanning of the abdomen and pelvis, the bladder is allowed to passively fill, either by having no urinary catheter and having the patient hold it, or by clamping the catheter if it is present. Unfortunately, this does not provide enough pressure to demonstrate small intraperitoneal bladder injuries and most extraperitoneal injuries.
The proper technique involves infusing contrast into the bladder through a urinary catheter. At least 350cc of dilute contrast solution must be instilled for proper distension and accurate diagnosis. This can be done prior to the abdominal scan. Once the initial scan has been obtained, the bladder must be emptied and a focused scan of just the bladder should be performed (post-void images). Several papers have shown that this technique is as accurate as conventional retrograde cystography, with 100% sensitivity and specificity for intraperitoneal ruptures. The sensitivity for extraperitoneal injury was slightly less at 93%.
Bottom line: Gross hematuria equals CT of the abdomen/pelvis and a proper CT cystogram, as described above. Don’t try to cheat and passively fill the bladder. You will miss about half of these injuries!
Related posts:
Reference: CT cystography with multiplanar reformation for suspected bladder rupture: experience in 234 cases. Am J Roentgenol 187(5):1296-302, 2006.

Intraperitoneal bladder rupture

Extraperitoneal bladder injury
Consequences of Using O+ Blood For Initial Transfusion In Trauma Patients
Blood transfusion is one of those potentially life-saving procedures that we take for granted in major trauma patients. Even the ATLS course recommends it for patients presenting in hemorrhagic shock. Unfortunately, it took quite a bit of work in the late 1800’s and early 1900’s to figure out how to keep from killing people when giving them someone else’s blood.
We now have very good systems for making sure that blood is safe, and that the appropriate antigens and antibodies are compatible in order to avoid serious transfusion reactions. Unfortunately, a lot of that goes out the door when a trauma professional needs blood now! A crossmatch takes about 45 minutes, and even making sure the major blood types match (ABO and Rh) takes about 15 minutes once you factor in paperwork and transport times.
All trauma centers now have access to “universal donor” blood. In the purest sense, this is O- blood, since it has no antigens that can activate the recipient’s immune system. However, O- comprises only 6.6% of the US blood supply, and it is frequently in short supply in smaller hospitals. An alternative is to use O+ blood, which is much more readily available (37.4% of the US supply).
But doesn’t this pose a risk of hemolytic transfusion reaction to patients who are Rh-? And what about women of childbearing age developing Rh antibodies that could cause hemolytic disease of the newborn? The Maryland Shocktrauma Center published a paper that showed some interesting results. It was a retrospective review of the 5,623 patients they saw in the year 2000 (!). A total of 480 of those patients received 5,203 units of blood.
Here are the interesting factoids:
- 161 received uncrossmatched O blood
- There were no acute transfusion reactions
- 14 patients lived to receive full crossmatch with Rh
- 4 females received O- and none developed antibodies
- Of the 10 males who received O+ blood, only 1 developed persistent Rh antibodies. One other had transient antibodies that disappeared at later testing.
Bottom line: Although it looks big, this is really just a small series. However, it does agree with other similar studies (listed in the bibliography of this paper). The use of uncrossmatched blood in general, and O+ blood specificially for males does appear to be safe. The low number of Rh antibody conversions may be due to the immunosuppression that goes along with both major trauma and blood transfusion. Consideration should still be given to administering RhoGam to males who are found to be Rh- after receiving Rh+ blood.
Related post:
Reference: Safety of Uncrossmatched Type-O Red Cells for Resuscitation from Hemorrhagic Shock. J Trauma 59(6):1445-1449, 2005.
