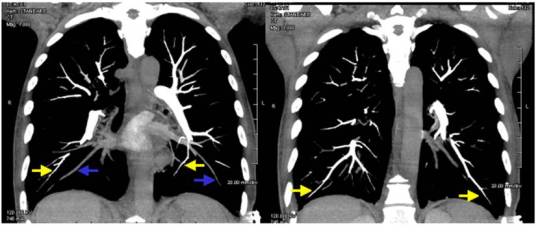
Fewer and fewer states have good helmet laws any more. Part of the problem is political. But the other part, may be… the end user. Here’s a piece written by a doctor and published in the St. Paul Pioneer Press in 1994 about a case he saw in medical school:
“I was working in a rural emergency room one day when the ambulance brought in a 17-year-old who had been in a motorcycle accident. It had just started raining, and the road was slick; he’d tried to take a corner too fast and had laid the bike down and skidded quite a long way.
“Fortunately, all he had were crapes and bruises. he had no head injury.
“Just as I was finishing up with him, a policeman came in and brought the young man his helmet, which they had taken off him at the scene. it was obviously a fancy and expensive helmet. It was a full-face unit – shiny, multicolored, metal-flake.
“One entire side of the helmet was ruined. It was deeply gouged and had obviously scraped along the asphalt with considerable impact for quite a distance. In some places, it appeared that the entire thickness of the hard shell had been penetrated, and you could see the soft inner lining.
“The young man was obviously very lucky.
“He looked at the helmet, groaned and said: ‘Oh, man! I just bought that a week ago! What a waste of $150!’”