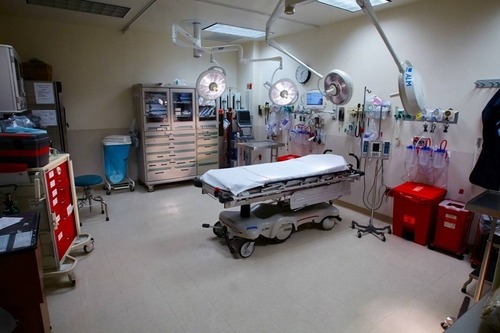
In the last two posts, I discussed the size of your trauma bay and how to measure it. This can obviously be helpful if you are updating or building new resuscitation rooms. But what about all the stuff that goes into it? Where is the best place to put it? If you are in the enviable position of being able to stock a brand-new room, here are some tips.
Figure out what you really need in the trauma bay. You don’t have to put everything and the kitchen stove in there. It’s fine to have less commonly used equipment somewhere else, but it must be close! You don’t want someone to have to walk 50 yards to look for something you need right now.
Here’s a list of the critical stuff:
- Temperature and light controls. These must be inside the room for easy and rapid access. And the doors should close to contain the heat. Resus rooms that are used frequently should be kept warm, doors closed, at all times.
- Personal protective equipment. This should be located just inside the room (if space is available) or just outside. It absolutely must be near the entrance and easily accessible or no one will wear it.
- Airway cart and video laryngoscope. These items must always be located near the head of the bed for immediate availability.
- Difficult airway cart. These are not used frequently, so need not be placed inside the room. But make sure it is close by.
- Travel ventilator. This can be stored outside unless you have lots of space available.
- IV start/blood draw carts. One of these should be stationed on either side of the patient.
- Rapid infuser. This may be located inside or outside of your trauma room based on the number of times it is typically needed.
- Procedure packs. These should be located inside the trauma bay, and clearly organized inside cabinets.
- Medication dispenser. This must be inside the room. Period.
- Other commonly used equipment/supplies. These should be placed intuitively in the bay and/or cabinets depending on frequency of usage of each item. Clear marking is essential.
- Scribe stand. Don’t forget the scribe. They obviously have to be in the room, and need some space for the (preferably) paper trauma flow sheet.
- Pediatric cart. This can be stored inside or outside the resus room, but should be nearby. Make sure that the measuring card that translates child size into equipment size is easily located.
- Blood refrigerator. This item is optional, but is becoming more common. It can be located inside or close outside the trauma bay depending on space available.
- Blanket and sheet warmer. These are nice to have, wherever you have room to put one. The patients will appreciate it.
- Procedure lights. Ceiling mounted are best because they don’t take up floor space. However, these are notorious for developing a mind of their own as they age. After a while, they never seem to stay focused on your field.
- Forced air warming blanket unit. This is important here in Minnesota, but also anywhere your patients can get cold. Which is pretty much everywhere. The airflow unit itself is relatively small and can usually be tucked under a counter somewhere. Otherwise, keep it nearby.
- Linens hamper. You need to get rid of that gown / those sheets and blankets / or whatever. There’s no reason to take up space in the room for this. Park it outside.
- Laundry basket. This is a valuable item that is generally overlooked. What do you usually do with all that stuff you cut off the patient? Drop it on the floor, right? This is setting you up to lose your patient’s stuff. Get a cheap plastic laundry basket from Target and put it under one of the counters. Toss clothing, shoes, etc in it as they are removed.
- Cast cart. These are typically huge. They can be anywhere else but inside the trauma bay. Roll it outside the door when needed.
Now where do you put all this stuff? Most trauma centers already have an established layout and flow in their existing trauma bays. When you are moving to a new one, plan ahead! Hopefully you will have more room, so you’ll have some additional flexibility as to where to place everything.
But designing the placement and flow on paper alone is of limited use. You must try it out in advance! How do you do this? Have your contractor mock up a space exactly the size of your new resuscitation room. Move actual carts, cabinets, and equipment into it. If it’s not possible to cart the exact stuff into it, have the contractor build mock-ups of them and place them in the bay.
Now have actual trauma team members practice simulations of common types of resuscitation: basic no frills, basic with intubation, basic with splinting/casting, advanced with all of the above plus multiple procedures. Take careful notes of flow and any glitches that arise. Then move your stuff around to fix any problems, and try again!