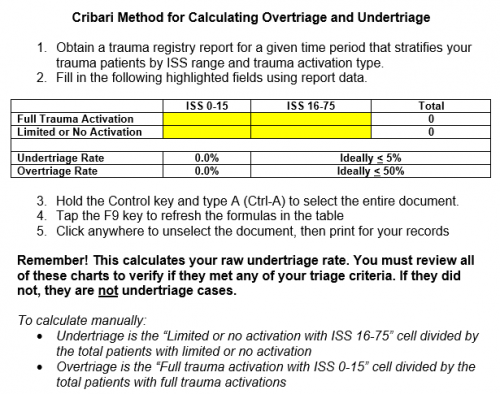
In my last post, I reviewed the use of the Cribari grid for evaluating over- and under-triage at your trauma center. This technique has been a mainstay for over a decade, but has its shortcomings. The most important one is that it relies only on the Injury Severity Score (ISS) to judge whether some type of mistriage occurred. As you know, the ISS is usually calculated after discharge, so it can only be applied after the fact.
Two years ago, the group at Baylor University in Dallas sought to develop an alternate method of determining who needed a full trauma team activation. They chose resource utilization as their surrogate to select these cases. They reviewed 2.5 years of their own registry data (Level I center). After several iterations, they settled on six “need for trauma intervention” (NFTI) criteria:
- blood transfusion within 4 hours of arrival
- discharge from ED to OR within 90 minutes of arrival
- discharge from ED to interventional radiology (IR)
- discharge from ED to ICU AND ICU length of stay at least 3 days
- require mechanical ventilation during the first 3 days, excluding anesthesia
- death within 60 hours of arrival
Patients who had at least one NFTI criterion were considered candidates for full trauma activation, and those who met none were not. Here are the factoids for this study:
- There were a total of 2260 full trauma activations and 2348 partial activations during the study period (a little over 900 per year for each level)
- Roughly 2/3 of full activations were NFTI +, and 1/3 were NFTI –
- For partial activations, 1/4 were NFTI + and 3/4 were NFTI –
- Only 13 of 561 deaths were NFTI – and all had DNR orders in place
The authors concluded that NFTI provides an assessment of both anatomy and physiology using only measures of early resource utilization. They believe that it self-adjusts for age, frailty, and comorbidities, and that it is a simple and effective tool for identifying major trauma patients.
Bottom line: This is an elegant attempt to improve upon the simple (yet admittedly flawed) Cribari matrix method for assessment of major trauma patient triage. It was thoughtfully designed and evaluated at this one center. The authors recognize that it is based on retrospective data, but so is the Cribari technique.
I believe that it may be an adjunct to Cribari. The matrix identifies gross under- and over-triage, but still requires the trauma program to review the outliers to see if mistriage actually occurred. It is basically a “first pass” that seeks to over-identify potential problem patients.
NFTI is similar, but it focuses on those patients who really should have been a full trauma activation due to their early need for critical resources to deal with their injuries. But is it enough? In my next post, I’ll review the follow-on paper from this group as they apply it to multiple trauma centers. And I’ll add some final thoughts on the subject.
Reference: Asking a Better Question: Development and Evaluation of the Need For Trauma Intervention (NFTI) Metric as a Novel Indicator of Major Trauma. J Trauma Nursing 24(3):150-157, 2017.