In my last post I discussed some of the basic effects of NSAIDs on bone healing. Now let’s see if theory applies to practice.
In 2003, several papers brought to light possible interactions between these drugs and fracture healing. Specifically, there were questions about these drugs interfering with the healing process and of increasing the number of delayed unions or nonunions. But once again, how convincing were these papers, really?
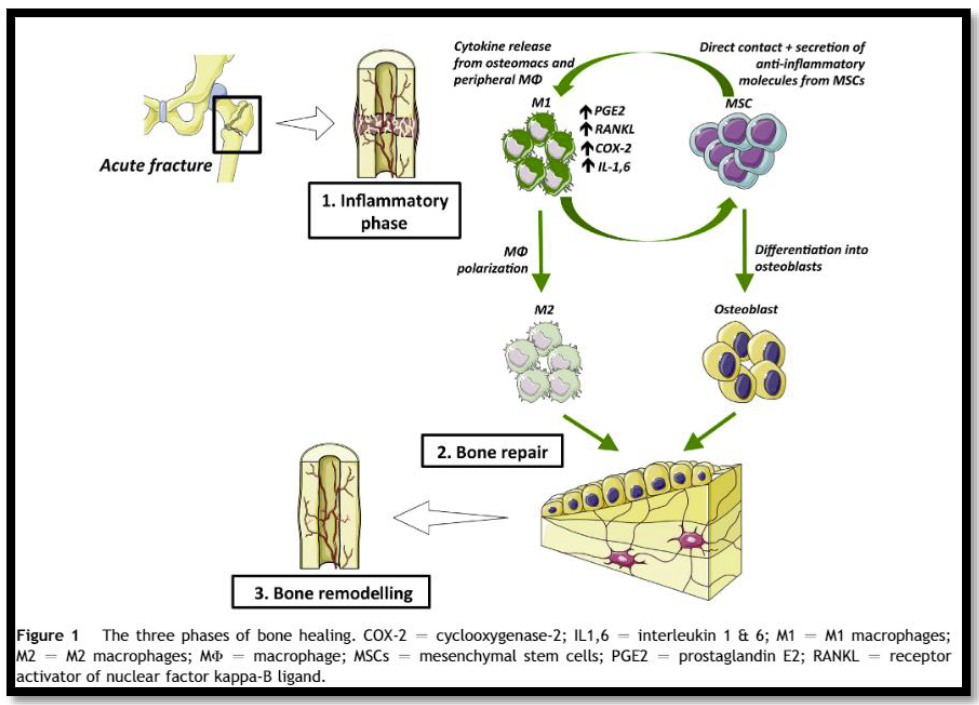
It would seem to make sense that NSAIDs could interfere with bone healing. The healing process relies heavily on the regulation of osteoblast and osteoclast function, which itself is regulated by prostaglandins. Since prostaglandins are synthesized by the COX enzymes, COX inhibitors like the NSAIDs should have the potential to impair this process. Indeed, animal studies in rats and rabbits seem to bear this out.
But as we have seen before, good animal studies don’t always translate well into human experience. Although a study from 2005 suggested that NSAID administration in older patients within 90 days of injury had a higher incidence of fracture nonunion, the study design was not a very good one. It was equally likely that patients who required these drugs in this age group may have been at higher risk for nonunion in the first place.
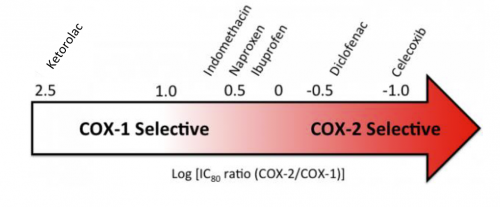
A meta-analysis of human studies was performed in 2011. Out of 558 potential studies, only 5 met criteria review. (This is yet another reminder of the sheer amount of sub-par research out there.) The authors found that short-term use (< 14 days) of normal dose NSAIDS was not associated with non-union. High doses of ketorolac (> 120mg/day) and diclofenac sodium (> 300mg total) did have an association. But remember, this does not show causation. There are many other factors that can impede healing (smoking, diabetes, etc).
A study from 2016 examined the effect of ketorolac administration on fracture healing in patients undergoing repairs of femoral and tibial fractures. It did not find an association between non-union and ketorolac, but did find one with smoking. Unfortunately, the study was small (85 patients given ketorolac, 243 controls without it). It probably does not have the statistical power to detect any difference with the NSAID. A power analysis was not provided in the methods section.
Bottom line: Once again, the animal data is clear and the human data less so. Although there are theoretical concerns about NSAID use and fracture healing, there is still not enough solid risk:benefit information to abandon short-term NSAID use in patients who really need them. NSAIDs can and should be prescribed in patients with short-term needs and simple fractures, and consider COX-1 specific drugs like ketorolac while your patient is in the hospital. And we do have some evidence that high-dose NSAIDs may have some impact, so stick to the usual doses for just as long as they are needed for pain management.
References:
- Effects of nonsteroidal anti-inflammatory drugs on bone formation and soft-tissue healing. J AM Acad Orthop Surg 12:139-43, 2004.
- Effect of COX-2 on fracture-healing in the rat femur. J Bone Joint Surg Am 86:116-123, 2004.
- Effects of perioperative anti-inflammatory and immunomodulating therapy on surgical wound healing. Pharmacotherapy 25:1566-1591, 2005.
- Pharmacological agents and impairment of fracture healing: what is the evidence? Injury 39:384-394, 2008.
- High dose nonsteroidal anti-inflammatory drugs compromise spinal fusion. Can J Anaesth 52:506-512, 2005.
- Nonsteroidal Anti-Inflammatory Drugs and Bone-Healing: A Systematic Review of Research Quality. JBJS Rev 4(3), 2016.
- High-dose ketorolac affects adult spinal fusion. Spine 36(7):E461-E468, 2011.
- Ketorolac administered in the recovery room for acute pain management does not affect healing rates of femoral and tibial fractures. J Orthop Trauma 30(9):479-482, 2016.